Regulator flags 90 unapproved drug combinations, asks states to act
In a significant intervention, India’s drug regulator has identified 90 fixed dose combination medicines that are being marketed without the required approval, and has asked state authorities to...
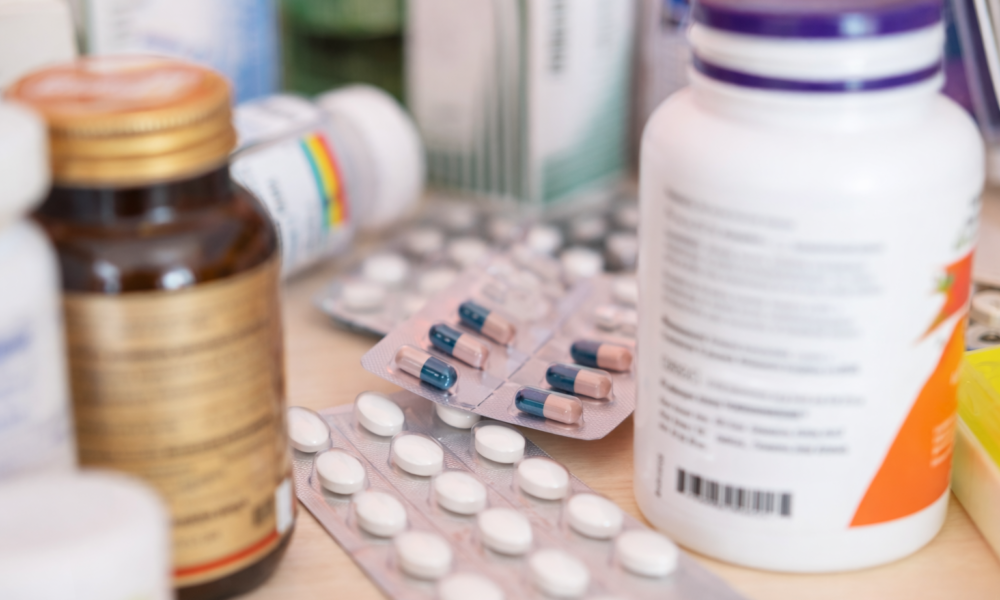
In a significant intervention, India’s drug regulator has identified 90 fixed dose combination medicines that are being marketed without the required approval, and has asked state authorities to initiate action against their manufacture and sale.
Fixed dose combinations, which blend two or more active ingredients into a single formulation, have long raised concerns when introduced without adequate clinical validation. The regulator’s latest communication underlines persistent gaps in compliance, despite earlier attempts to streamline approvals and curb irrational combinations.
The directive places the onus on state drug controllers to review licences, halt distribution where necessary, and ensure that manufacturers adhere to established safety norms. It also signals a renewed push by the Centre to tighten oversight in a fragmented regulatory landscape where enforcement often varies across states.
For patients, the move is a reminder of the risks posed by unverified therapies that may offer convenience but lack proven efficacy or safety. For the pharmaceutical industry, it is another indication that regulatory scrutiny is unlikely to ease, particularly in segments where oversight has historically been uneven.











No Comment! Be the first one.