Weight-loss drugs under scrutiny as regulators move to curb misuse
The Centre’s decision to tighten oversight of GLP-1 based weight-loss drugs comes at a time when demand for these therapies is rising sharply beyond their approved medical use. Originally indicated...
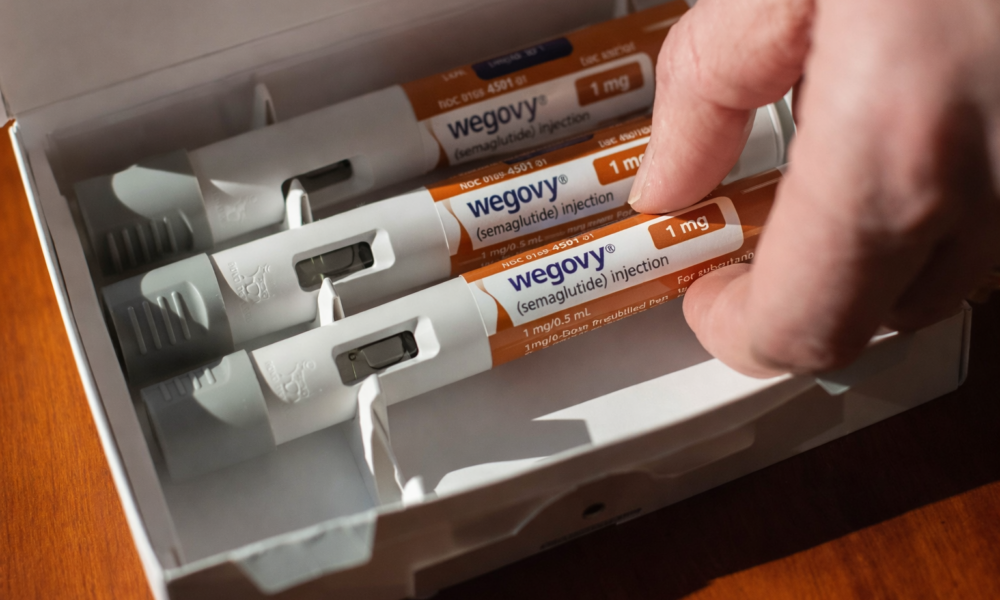
The Centre’s decision to tighten oversight of GLP-1 based weight-loss drugs comes at a time when demand for these therapies is rising sharply beyond their approved medical use. Originally indicated for diabetes and, in specific cases, obesity under clinical supervision, these drugs are increasingly being marketed as quick fixes for weight reduction, often outside regulated channels.
Regulators are now seeking to rein in unauthorised sales and promotional practices that blur the line between prescription therapy and lifestyle product. The concern is not merely about misuse, but about the absence of medical oversight in a category of drugs that can have significant side effects if used indiscriminately.
The popularity of these medications has been fuelled by social media trends and word-of-mouth endorsements, creating a parallel market that operates with limited accountability. In many instances, patients are accessing these drugs without adequate screening for underlying conditions or without guidance on dosage and duration.
This has prompted authorities to step up surveillance across the supply chain, including pharmacies and online platforms. The move also signals a broader regulatory challenge. As newer therapies enter the market and public awareness grows, the risk of off-label use and commercial exploitation increases.
At its core, the issue underscores the need to reinforce the distinction between medically necessary treatment and consumer driven demand. Without stricter enforcement and clearer communication, the misuse of such drugs could evolve into a larger public health concern rather than a controlled therapeutic intervention.













No Comment! Be the first one.